
Barrier Films: High-Performance Polymers
Our specially formulated films protect products against moisture, gas, oxygen, and UV rays, providing maximum efficacy and increased shelf life.
The Philosophy
Packaging Is Protection.
In regulated industries, packaging is not decoration. It is science. It is barrier performance. It is compliance discipline. It is brand trust in physical form. We engineer flexible packaging systems that perform under pressure, scale with demand, and comply with global standards.

Built by Solution
Triplex Barrier Films
Multi-layer blister structures combining PVC, PE, and PVdC layers for enhanced moisture and oxygen protection.
- Enhanced moisture barrier
- Improved oxygen protection
- Strong thermoforming performance
- High mechanical stability
- Suitable for moisture-sensitive formulations
- Available in multiple specifications and colours
- PVC
- PVC / PE
- PVC / PVdC
- PVC / PE / PVdC (Triplex Structure)
- PVC thickness: 250 µm – 350 µm
- PE thickness: 25 µm – 30 µm
- PVdC coating: 40 g/m² – 120 g/m²
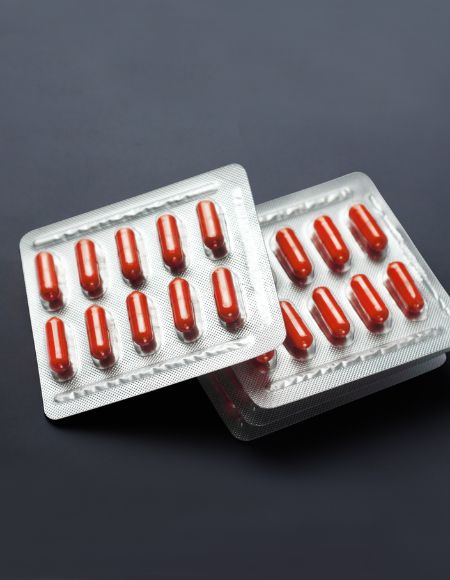

Aclar® Laminates
High-barrier structures offering superior moisture protection and chemical stability for highly sensitive formulations.
- Superior moisture barrier
- Ideal for moisture-sensitive drugs
- Chemically inert material
- Compatible with tablets, capsules, and soft gels
- Clear thermoformable structure
- Optimized barrier thickness options
- PVC / Aclar
- PVC / PE / Aclar
- PVC / PE / EVOH / PE / Aclar
- PVC / Aclar / PVC
- APET / Aclar
- APET / PE / EVOH / PE / Aclar
- PVC thickness: 100 µm – 450 µm
- Aclar thickness: 15 µm – 152 µm
- PE thickness: 25 µm – 50 µm
- PE / EVOH / PE barrier layer: 70 µm
- APET thickness: 200 µm – 450 µm
Duplex Barrier Films
: PVC–PVdC coated materials providing moderate to high moisture and oxygen protection.
- Moderate to high moisture barrier
- Effective oxygen protection
- Reliable thermoforming performance
- Suitable for pharmaceutical blister packs
- Available in multiple coating grades
- Offered in a range of colours and specifications
- PVC / PVdC (Duplex Structure)
- PVC thickness: 200 µm – 400 µm
- PVdC coating: 40 gsm – 120 gsm
- Available coating grades: 40 / 60 / 80 / 90 / 120 gsm


Rigid & Aluminium-Look PVC Films
Cost-effective, highly thermoformable films available in clear, colored, or high-impact metallic aesthetics.
- Reliable thermoforming performance
- Cost-effective blister packaging material
- Suitable for stable drug formulations
- Compatible with tablets and capsules
- Available in multiple colours and grades
- Suitable for high-speed blister packing lines
- PVC
- PVC thickness: 200 µm – 400 µm
- Standard thickness: 250 µm
- Available in transparent and coloured variants
Anti-Counterfeit Foils
Specialized materials integrating embedded visual security elements within the thermoformable structure.
- Metallic shelf-impact appearance
- Excellent blister formability
- Ideal for brand differentiation
- Compatible with high-speed blister lines
- Available in vibrant metallic colours
- Optional high-barrier coating variants
Regular Structures
- PVC
- PVC / PVdC
- PVC / PE / PVdC
High Barrier Structures
- PVC / PVdC Ultrasafe
- PVC / PE / PVdC Ultrasafe
- PVC thickness: 100 µm – 450 µm
- Standard PVC thickness: 250 µm
- PVdC coating grades: 40 / 60 / 80 / 90 / 120 gsm
- PVdC Ultrasafe coating: 90 gsm / 120 gsm
- PE thickness: 25 µm – 30 µm

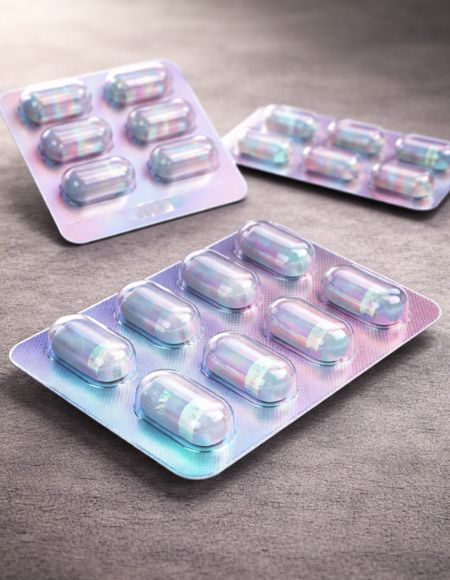
Anti-Counterfeit Films
Anti-counterfeit films are specialized blister packaging materials designed to enhance product authentication and protect pharmaceutical brands from duplication or tampering. PSI offers anti-counterfeit blister film solutions through qualified global partners, integrating embedded visual security elements within thermoformable blister structures to support brand protection and packaging differentiation.
- Integrated anti-counterfeit visual features
- Embedded image security technology
- Enhances brand authentication
- Supports product differentiation on shelf
- Compatible with standard blister packaging lines
- Available in clear, coloured, or metallic formats
- PVC
- PVC / PVdC
- PVC thickness: 200 µm – 400 µm
- Standard PVC thickness: 250 µm
- PVdC coating grades: 40 / 60 / 80 / 90 / 120 gsm
Unit Dose Films
Designed for single-use liquid and semi-liquid product packaging.
- Designed for single-dose packaging
- Suitable for liquid and semi-liquid formulations
- Provides moisture and oxygen protection
- Compatible with unit-dose packaging machines
- Ideal for samples and trial packs
- Supports surface and reverse printing options
- PVC / PE
- PVC / PVdC / PE
- PVC thickness: 200 µm – 400 µm
- PE thickness: 30 µm – 50 µm
- PVdC coating: 40 gsm


Suppository Laminates Films
Suppository laminate films are specialized packaging structures designed for thermoforming and sealing suppository doses while maintaining product stability and protection. PSI provides suppository laminate film solutions through qualified global partners, enabling reliable forming performance, barrier protection, and compatibility with pharmaceutical suppository packaging lines.
- Designed for suppository dose packaging
- Moisture and oxygen protection
- Reliable thermoforming performance
- Compatible with suppository packaging machines
- Available in translucent, white, and opaque variants
- Supports surface printing options
- PVC / PE
- PVC / PVdC / PE
- Alu / PE
- PVC thickness: 100 µm – 130 µm
- PVdC coating: 40 gsm
- Aluminium layer: 45 µm
- PE thickness: 30 µm – 40 µm

WHY CHOOSE US
Consistency At Scale.
Engineered through deep technical understanding of packaging substrates. Customized by application, process requirements, and end-use conditions. Aligned with global compliance standards where consistency is non-negotiable.
- Technical substrate engineering
- Application-driven customization
- Global compliance alignment
- When failure is not an option, discipline matters.
Where Performance Meets Reliability
From barrier films to labels and cartons, our customers trust us to deliver packaging that protects products and supports compliance.
An orci nullam tempor sapien, eget orci gravida donec enim ipsum porta justo integer at odio velna auctor. Magna undo ipsum vitae purus ipsum primis in laoreet augue lectus
Sapien sem accumsan vitae purus diam integer congue magna undo. Magna, sodales vitae aenean mauris tempor at risus lectus aenean magna and mauris lectus laoreet tempor
Etiam sapien sem accumsan sagittis congue. Suscipit and egestas magna sodales vitae aenean mauris tempor risus lectus aenean aenean mauris laoreet tempor
Egestas egestas magna ipsum vitae purus efficitur ipsum in primis cubilia laoreet augue congue. Suscipit sodales vitae lobortis magna donec enim
An orci nullam tempor sapien, eget orci gravida donec enim ipsum porta justo integer at odio velna auctor. Magna undo ipsum vitae purus ipsum primis in laoreet augue lectus
Mauris donec ociis et magnis sapien etiam sapien sagittis congue augue. An orci nullam tempor sapien, eget gravida a donec enim ipsum porta
Egestas egestas magna ipsum vitae purus efficitur ipsum in primis cubilia laoreet augue congue. Suscipit sodales vitae lobortis magna donec enim
Let’s Build the Right Packaging System.
Share your application, regulatory requirements, and performance expectations. We will engineer a solution aligned to your technical and commercial goals.






